Our Institute
Clinical Trials
Our Science
News
International Patients
- International Patients
- International patient Service Care
- Travel Arrangement and Hospital Admission
- FAQ
- Contact Us
In the past decade, the US FDA has approved many tumor immunotherapy products. The application of CAR-T therapy in solid tumors has proven safe yet with limited anti-tumor efficacy. The average overall survival period is less than 1 year, and most patients still relapse with progressive disease.
Although CD19 CAR-T therapy has proven highly effective in treating B cell malignancies, there are still many challenges in CAR-T therapy for solid tumors including: what tumor antigens to target, how to avoid antigen loss, and how to prevent the exhaustion of CAR-Ts in the body.
Besides the target antigen escape, the heterogeneity and diversity of the tumor-associated antigens plus the suppressive tumor microenvironment are known difficulties in CAR-T therapy targeting solid tumors.
In treating B cell malignancies, a single dose of CD19-targeted CAR-T cells after Cy/Flu chemotherapy conditioning can persist at low frequencies for several months, which may be maintained by sustained exposure to the existing CD19-positive B cells. These factors contribute to the long-lasting remission in many patients treated with the CD19-CAR-T cells. However, in solid tumor CAR-T therapies, there are still many challenges different from treating blood cancers.
Recently, a team from Baylor College of Medicine presented a multiple CAR-T infusion strategy for the treatment of recurrent and refractory sarcoma, and documented the course of response in a child with refractory bone marrow metastatic rhabdomyosarcoma treated with autologous HER2 CAR-T cells.

【10 rounds of CAR-T treatment. The first three times of CAR-T infusions were combined with chemotherapy pretreatment to promote the expansion of HER2 CAR-T cells. After remission (CR) was obtained, no pretreatment was performed, and four infusions of CAR-Ts were added for consolidation. However, after stopping the treatment for 6 months, the disease recurred in the bone marrow. At this time, after pretreatment, CAR-Ts were infused again and CR was achieved. After that, two more rounds of chemo-conditioning plus CAR-T were carried out for consolidation, which was combined with PD-1 immune blocking antibody for a synergistic effect (injected every 2-3 weeks), and thereby CR had been maintained for more than 1 year.】
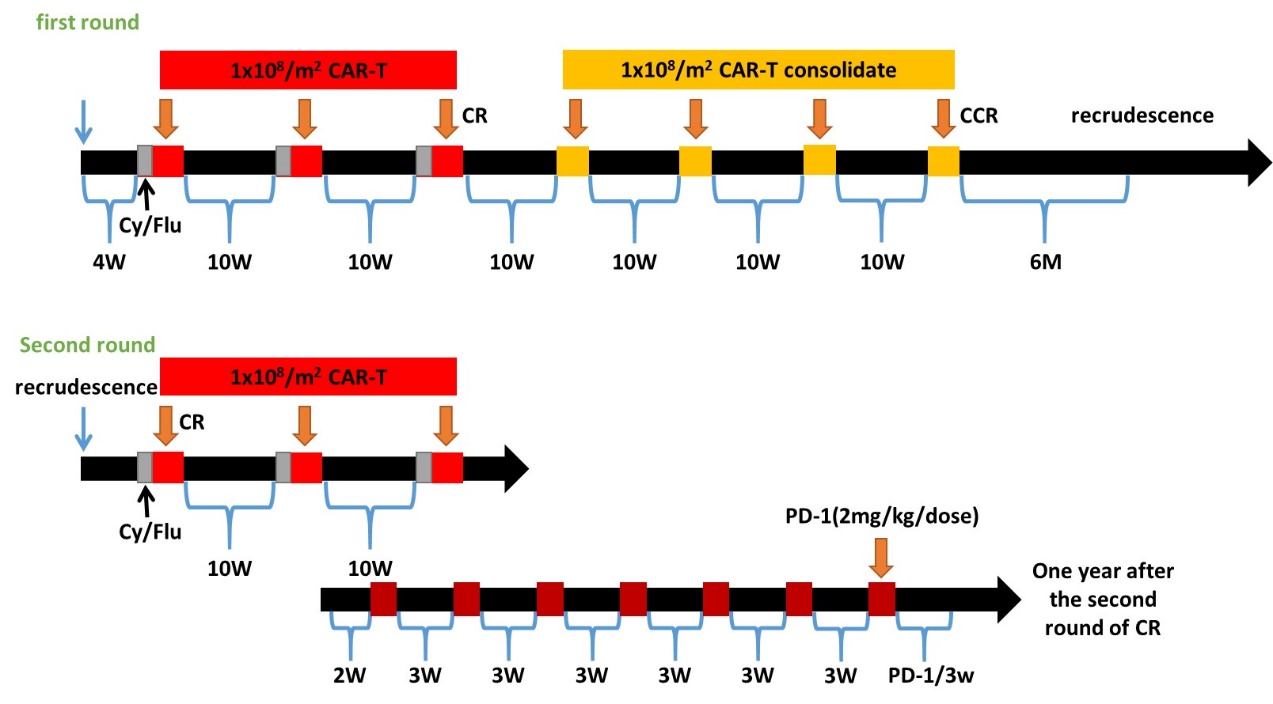
Lessons we learned
After cancer relapse, the original targeted tumor antigens may disappear. For leukemia and lymphomas, GIMI has launched the "4SCAR2.0" treatment strategy since 2016: it is based on infusions of multiple tumor-specific CAR-Ts for consolidation and maintenance, which improves the long term survival of patients. Multiple CAR-T combinations effectively overcome tumor mutations and antigen escape, while prolong the CAR-T persistence. The 4SCAR2.0 strategy includes a primary CAR-T treatment, a booster CAR-T infusion, and subsequent consolidation CAR-T infusions to maintain a stable level of tumor-specific CAR-T cells in the body to prevent recurrence.
The treatment of solid tumors with CAR-T can be more difficult than the treatment of blood cancer. Multi-target CAR-T infusions will be the key to a long-term remission. The above Baylor study using 10 rounds of CAR-T infusions for sarcoma treatment is basically not translatable in real-world clinical practice. However, GIMI has overcome this challenge and made the highly individualized treatment schema doable and affordable to the general public.
GIMI medinfo 12.2022
By CW Chang